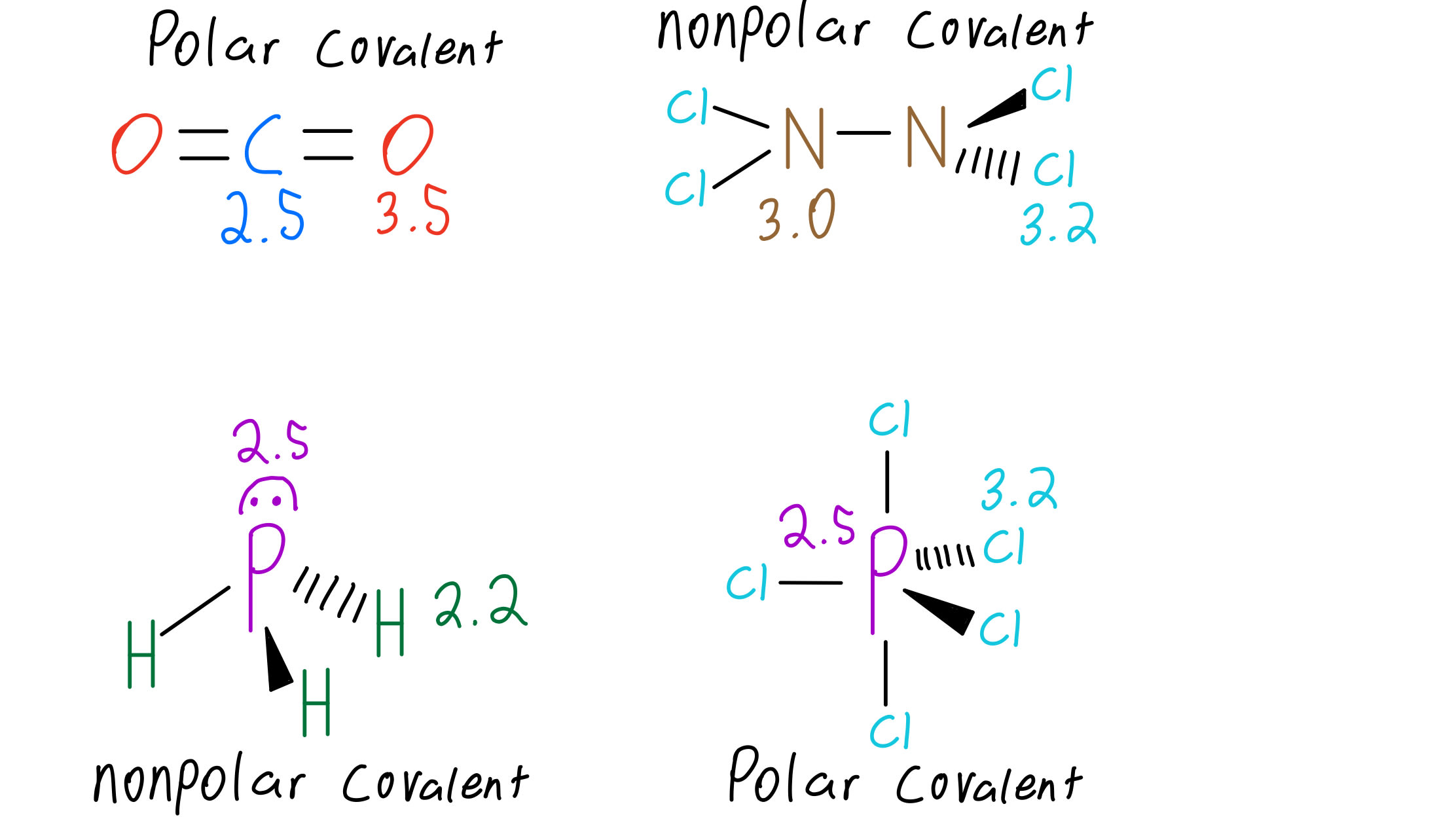By drawing the Lewis structures of the following molecules, determine which of them is polar. a. Cl2 b. Bh3 c. NH3 d. PCl5 | Homework.Study.com

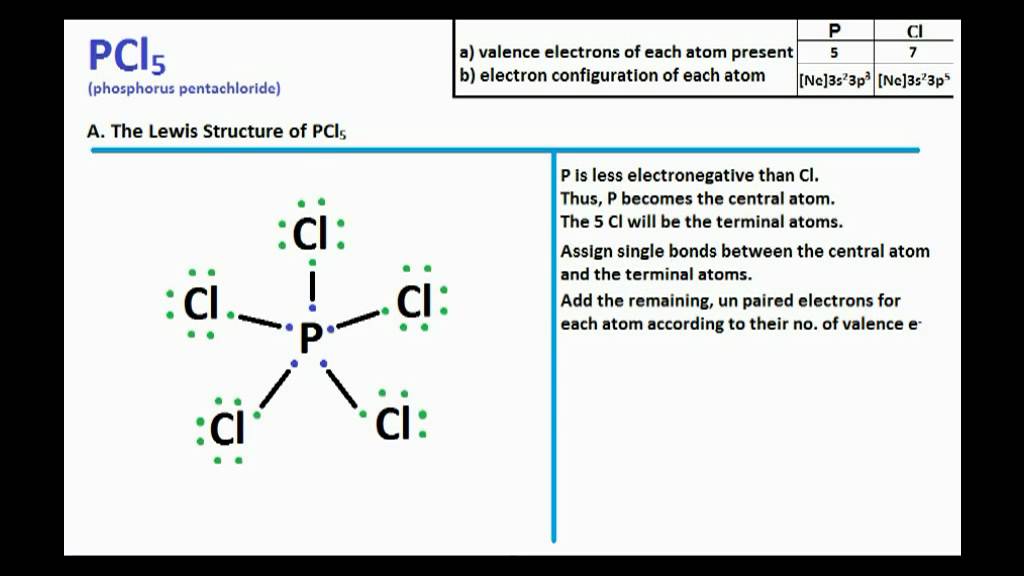
PCl5 Lewis Structure: Drawings, Hybridization, Shape, Charges, Pair And Detailed Facts – Lambda Geeks

Draw the Lewis structures of the molecules below and use them to answer the following questions: I. - Brainly.com

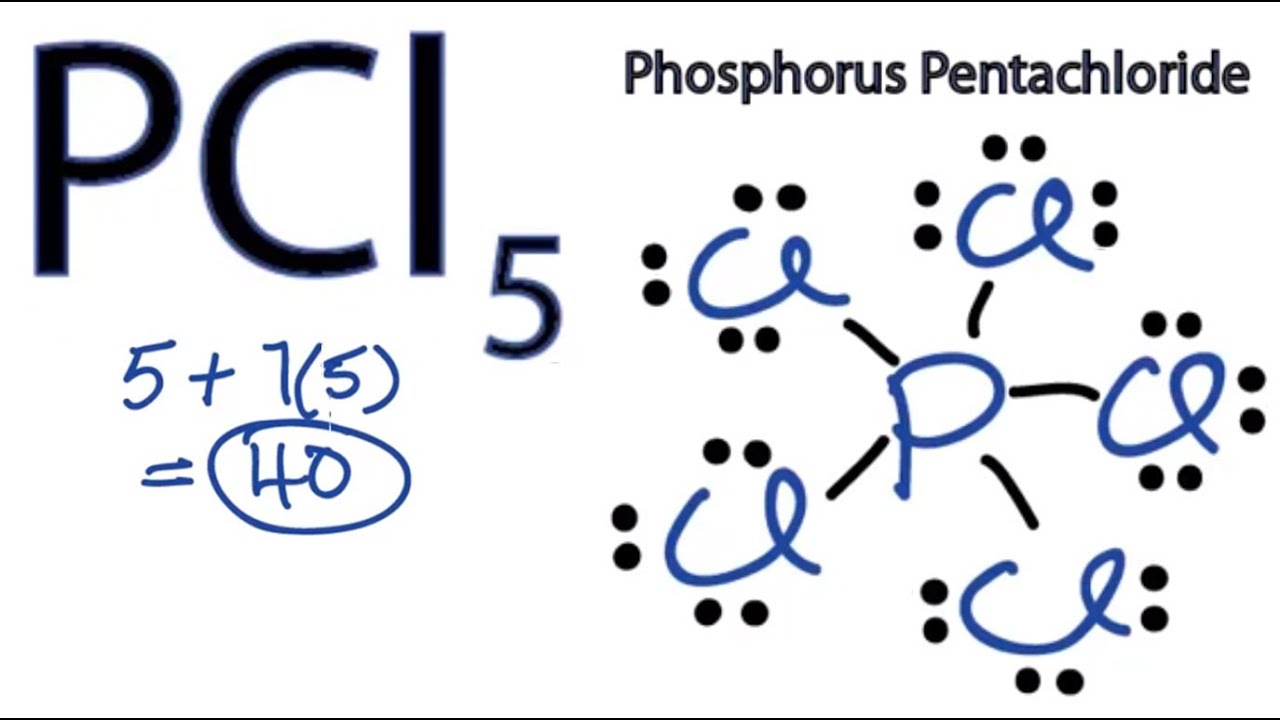
Draw the Lewis structure for PCl5 and answer the following questions. a. Does the central atom violate the octet rule? b. How many lone pairs of electrons are in the molecule? c.

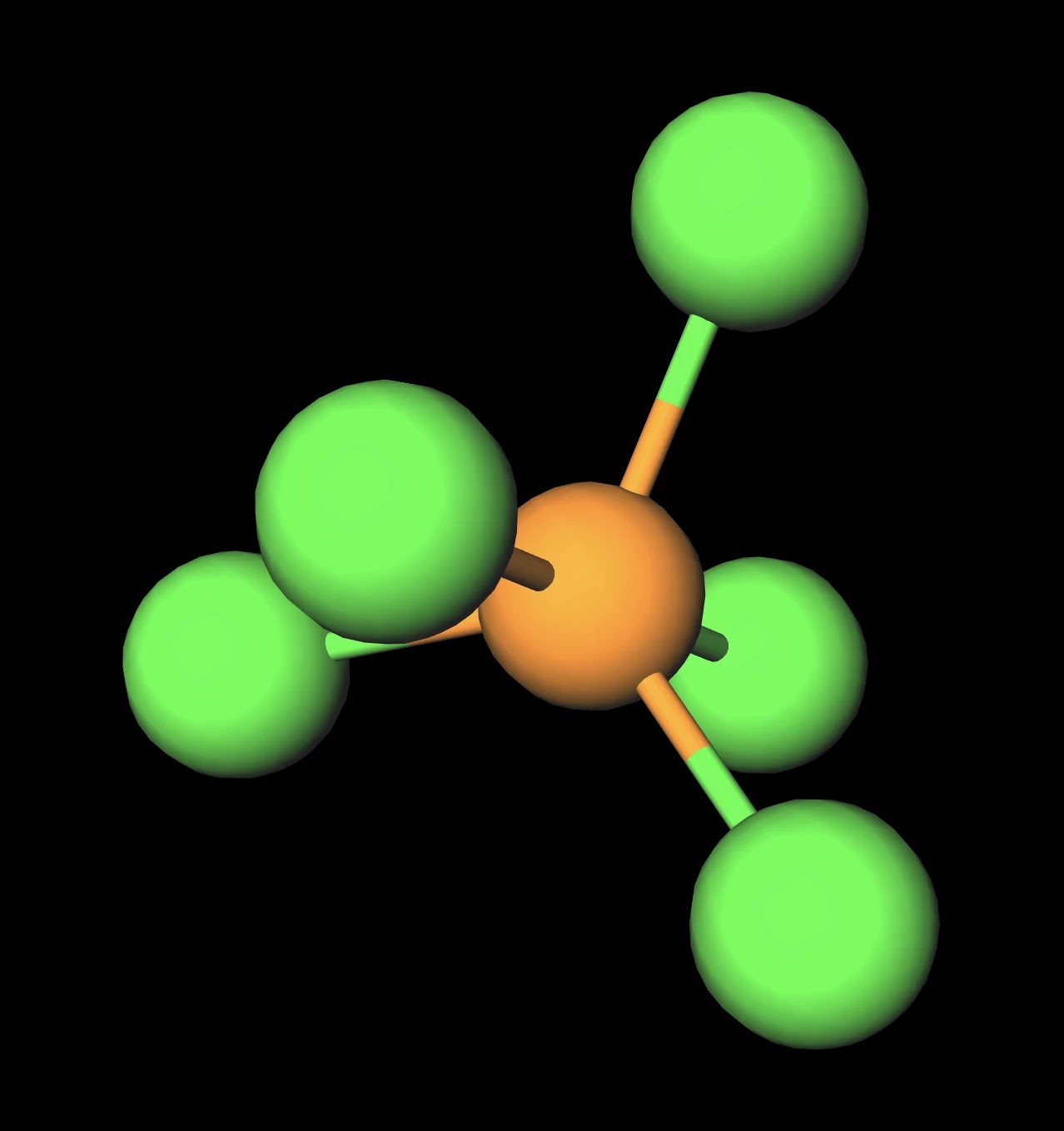
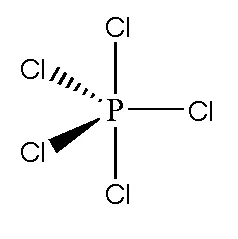
For the molecule PCl5 (phosphorus pentachloride), draw a proper Lewis structure that clearly identifies all electrons and has the predicted geometric (VSEPR) shape. Name and comment on the polarity of the molecule (

PCL5 Lewis Structure (Phosphorous Pentachloride) | PCL5 Lewis Structure (Phosphorous Pentachloride) Hi Everyone! Welcome back to our channel. In today's video, we help you determine the Lewis Structure... | By Geometry Of